Sterilization Packaging Materials — Sterilization Wrap, Non-woven Fabric, Crepe Paper, Paper-Plastic Pouches, and the Establishment of a Sterile Barrier
Sterilization Packaging Materials — Sterilization Wrap, Non-woven Fabric, Crepe Paper, Paper-Plastic Pouches, and the Establishment of a Sterile Barrier


ISO 11607-1:2006, Packaging for terminally sterilized medical devices – Part 1: Requirements for materials, sterile barrier systems, and packaging systems, explicitly outlines specific requirements for materials, sterile barrier systems (SBS), and packaging systems. Concurrently, the EN 868 series (EN 868-2 through EN 868-10), which covers packaging materials and systems for medical devices to be sterilized, also provides comprehensive specifications for medical device packaging.
Currently, ISO 11607 and EN 868 are internationally recognized as the definitive standard frameworks governing the packaging of terminally sterilized medical devices.
Fundamental Concepts of Packaging
After harmonizing medical device packaging terminology across various nations, ISO 11607-1 adopts the term "Sterile Barrier System" to describe the primary packaging for terminally sterilized medical devices. Products such as paper pouches and paper-plastic combination pouches used for medical device packaging, manufactured by Nanjing Sengong Biotechnology Co., Ltd., are classified as "Preformed Sterile Barrier Systems."
As a special case, packaging designed for sterile fluid paths—where sterility of the components intended to contact liquids is ensured by the sealing integrity of protective sleeves and other closure devices—is also considered a Sterile Barrier System.
A packaging system typically consists of two main components: the Sterile Barrier System (SBS) and Protective Packaging. The Sterile Barrier System provides the fundamental sterile barrier for terminally sterilized medical devices, within which the devices undergo sterilization. Protective Packaging serves as secondary packaging, offering additional protection to the Sterile Barrier System.
Currently, common Sterile Barrier Systems are categorized into reusable and single-use systems.
Reusable Sterile Barrier Systems include reusable woven textiles (surgical linens) and sterilization instrument containers (rigid containers).
Single-Use Sterile Barrier Systems encompass disposable sterilization materials such as nonwovens, creped paper, and paper-plastic pouches.
The following section provides an overview of several specific material configurations:
Preformed Tray and Lid Combination Packaging
In this packaging configuration, the tray is typically preformed using thermoforming or compression molding processes. The lid, which has a fixed shape, may be either breathable (porous) or non-breathable (non-porous). Typical lids feature a heat-seal coating that allows them to be heat-sealed directly onto the tray. This packaging format is generally used for larger, heavier instruments, such as orthopedic implants, pacemakers, and surgical instrument setsFlexible Peelable Combination Pouches: The typical structure of a combination pouch consists of a film on one side and a film, paper, or nonwoven material on the other. These pouches are commonly supplied as Preformed Sterile Barrier Systems. All seals are pre-formed except for one opening (typically at the bottom), which remains open to allow instrument insertion before final sealing. Since combination pouches can be manufactured in various widths, they serve as the Sterile Barrier System for a wide range of instruments, particularly those characterized by small size and light weight.
These pouches feature various design options. For instance, they can be constructed as gusseted pouches (also known as 3D pouches), which are suitable for thicker instruments, or as flat pouches. The film layer allows for visual inspection of the contents prior to opening, while the paper layer facilitates the penetration of sterilizing agents (such as steam or ethylene oxide gas) into the package.
Sterilization Paper Pouches: Sterilization paper pouches are manufactured exclusively from medical-grade porous paper, folded into an elongated tubular shape. The tube is sealed along its length with a double line of adhesive and then cut to the required specifications. One end is sealed with one or more layers of adhesive and folded multiple times to enhance seal integrity. The open end is typically designed with an offset edge or a thumb notch to facilitate opening the pouch during instrument loading. The final closure of the pouch is completed prior to sterilization.
Form-Fill-Seal (FFS) Process Packaging: This Sterile Barrier System is manufactured via an integrated three-in-one production line (automation). It is available in combination pouch formats, preformed tray-and-lid configurations, or as a shape formed from a flexible bottom film. During the FFS process, the top and bottom packaging components are loaded into the FFS machine. The machine's operational sequence involves forming the bottom component, loading the instruments, applying the top component, and finally sealing the unit to create the Sterile Barrier System.
Specialized Medical Nonwoven Wraps: Distinct from standard nonwovens, these are utilized for sterilization packaging due to their high tensile strength, superior breathability, and effective bacterial barrier properties. Since the 1980s, they have demonstrated excellent performance in infection control applications, effectively addressing the limitations of other packaging materials. Additionally, high-performance creped paper remains a viable and effective choice for sterilization packaging.
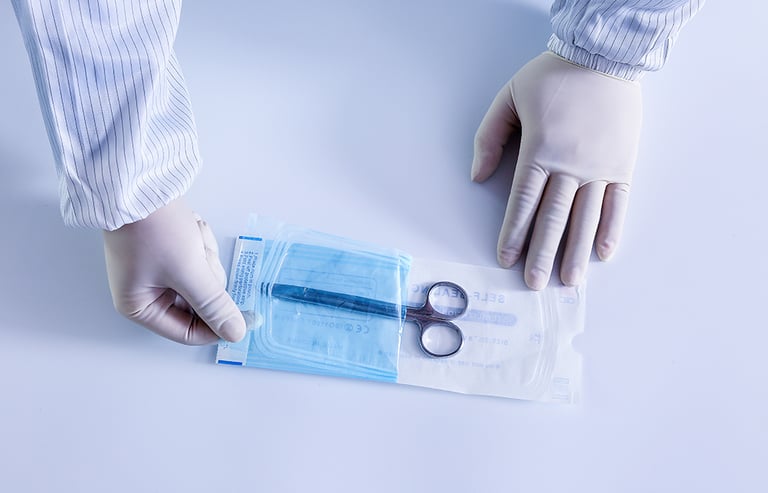


The sections above provide a detailed overview of sterilization packaging products. Sainsmed supplies the full range of these sterilization packaging solutions. All products have undergone rigorous testing and comply with ISO 13485, CE, ISO 11607, EN 868, and ISO 10993 standards. With extensive manufacturing experience in materials used within Central Sterile Supply Departments (CSSD), Sainsmed offers customized solutions tailored to specific customer requirements.
Office Location:
Medmoist Inc Corporation
1500 N GRANT ST, STE R, DENVER, CO 80203, US
Branch Offices:
22 F.-3, No. 285, Sec. 2, Taiwan Blvd., Minlong Vil., West Dist., Taichung City 403502, Taiwan
Contact Information:
Sales@sainsmed.com
Copyright © 2026 Sainsmed. All Rights Reserved. Sainsmed is a brand owned and operated by Medmoist Inc.
