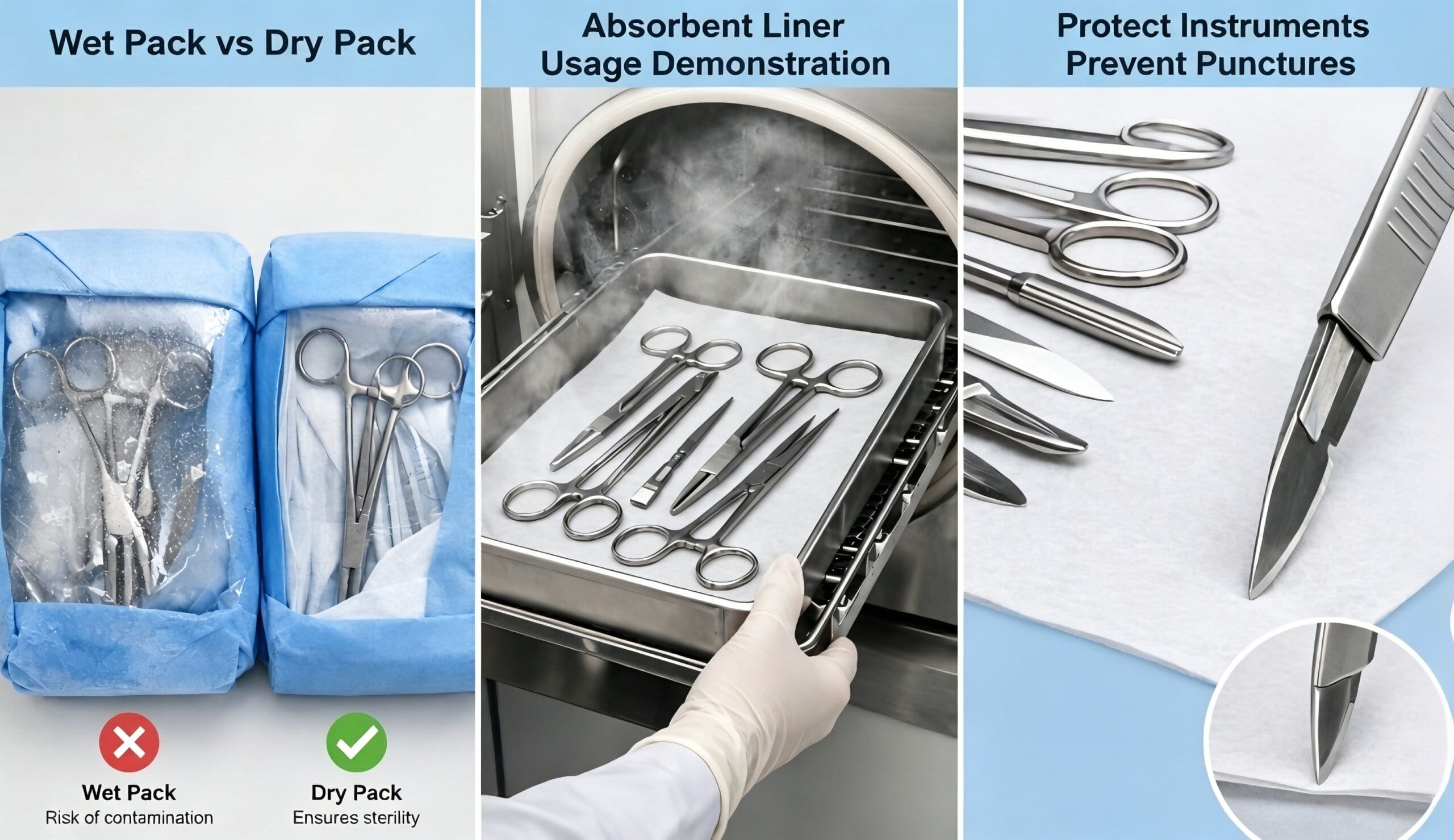
Addressing wet pack challenges with absorbent liner solutions in clinical sterilization workflows
Understanding the Clinical Risk of Wet Packs
Wet packs, defined as moisture present on or within sterilized packages after steam sterilization, represent a significant breach in sterility assurance. According to guidelines from AAMI and CDC, any sterile package found wet must be considered contaminated and requires reprocessing. Moisture facilitates microbial migration through capillary action, increasing the risk of surgical site infections (SSI). This issue is especially critical in Central Sterile Supply Departments (CSSD), where packaging integrity directly impacts patient safety and compliance with infection control standards.
Root Causes of Wet Pack Formation
Wet packs arise from multiple factors, including excessive load density, insufficient drying time, poor steam quality, and condensation during cooling. In addition, inadequate internal moisture management—such as the absence of absorbent materials within instrument trays—can lead to water accumulation. Clinical evidence indicates that complex surgical instruments, especially those with lumens or heavy metal mass, are more prone to retaining residual moisture, making effective drying more challenging.
Absorbent Liner Pads: Mechanism and Clinical Value
Absorbent liner pads are designed to be placed at the bottom of instrument trays to actively manage moisture during sterilization cycles. Based on the provided documentation, these pads are manufactured using high-absorbency medical-grade materials that rapidly capture condensate generated during steam sterilization. By stabilizing the internal microenvironment of the sterile barrier system, they prevent moisture from migrating դեպի outer packaging layers, thereby preserving sterility.
Recent clinical practices and CSSD optimization studies (2023–2025) demonstrate that the use of absorbent liners can significantly reduce wet pack incidence, particularly in high-volume hospital settings. Their integration supports compliance with international sterilization standards and enhances overall process reliability.
Clinical Application and Workflow Integration
Absorbent liner pads are applicable across multiple surgical disciplines, including orthopedics, general surgery, and neurosurgery. In routine practice, they are placed beneath surgical instruments inside trays or rigid sterilization containers prior to autoclaving. This is particularly beneficial for heavy instrument sets or high-density loads, where condensation risk is elevated.
From an operational standpoint, incorporating absorbent liners into CSSD workflows reduces the frequency of re-sterilization caused by wet packs. This leads to improved turnaround efficiency, reduced labor burden, and decreased wear on surgical instruments. For healthcare institutions, this translates into better resource utilization and improved surgical scheduling reliability.
Addressing Industry Pain Points and Ensuring Compliance
Wet pack management remains a persistent challenge for healthcare providers aiming to meet strict infection control requirements. Absorbent liner pads directly address key pain points, including sterilization failure, resource waste, and workflow inefficiencies. They offer a simple yet effective intervention without requiring significant changes to existing sterilization equipment or protocols.
As healthcare systems increasingly prioritize quality assurance and cost control, solutions that enhance sterilization outcomes while maintaining operational efficiency are essential. Absorbent liners provide a practical, scalable approach to improving CSSD performance and ensuring compliance with global standards.
Conclusion
Wet packs pose a critical risk to sterility and patient safety in medical environments. Effective management requires both optimized sterilization parameters and reliable moisture control strategies. Absorbent liner pads serve as an evidence-based solution, significantly improving drying performance, reducing contamination risk, and enhancing workflow efficiency. Their adoption is a key step toward achieving safer and more consistent sterilization outcomes in modern healthcare systems.