Evaluating wet pack risks and compliance with FDA and EU MDR requirements in CSSD practice
Structural Characteristics of Sterilization Containers and Wet Pack Risks
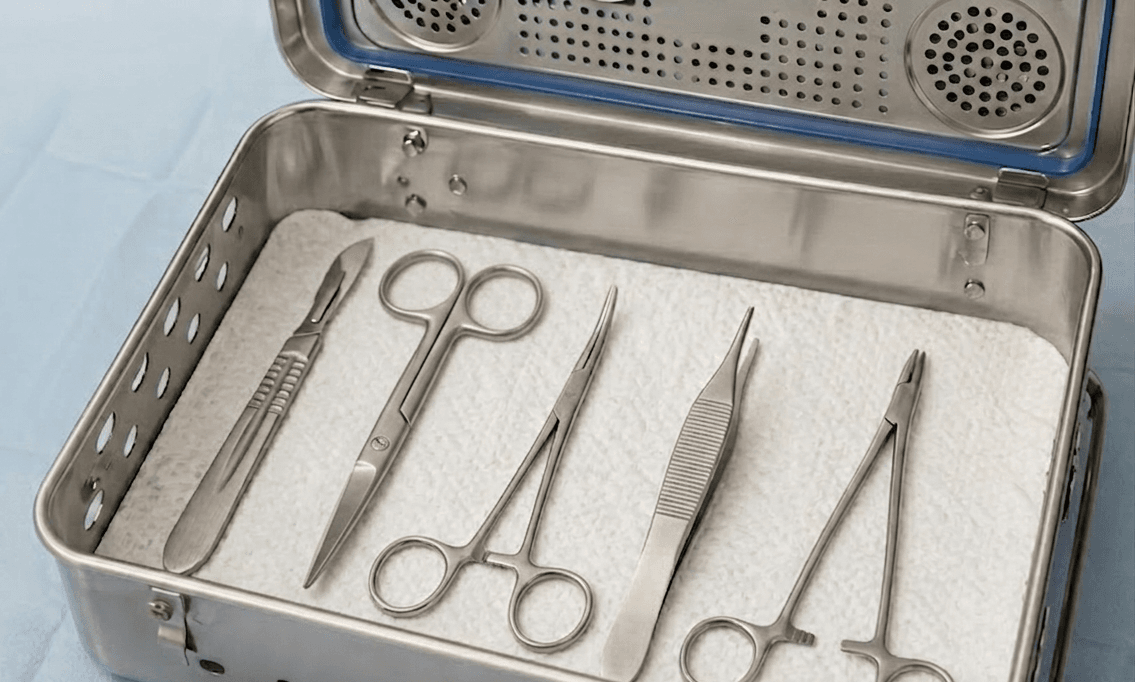
Sterilization container systems are widely used in Central Sterile Supply Departments (CSSD) and operating rooms as an alternative to traditional wrapping methods. Their rigid structure improves workflow efficiency and reduces packaging variability. However, unlike wrapped trays, these containers rely on limited filter or valve systems—typically circular sterilization ports—for steam دخول and condensate discharge.
In clinical practice, this structural limitation can restrict effective drainage of condensate during steam sterilization cycles. When residual moisture is not fully eliminated during the drying phase, it can result in the formation of “wet packs.” Compared to wrapped instrument sets, containerized systems inherently carry a higher risk of moisture retention due to constrained airflow and drainage pathways. This not only compromises the sterile barrier integrity but may also necessitate complete reprocessing of the instrument set, increasing operational costs and infection risks.

Role of Absorbent Liners in Moisture Control
To address these limitations, the placement of absorbent liner pads inside sterilization containers has become a critical intervention for moisture management. Positioned at the bottom of the container, these liners actively absorb condensate generated during sterilization and cooling phases.
Recent CSSD practice guidelines and infection control recommendations highlight several key benefits of absorbent liners: reducing moisture accumulation at the base of instrument trays, improving drying efficiency, decreasing the incidence of wet packs, and maintaining post-sterilization sterility stability. These liners are typically manufactured from medical-grade cellulose or composite fibers, offering high absorbency while maintaining steam permeability.
Importantly, their use does not interfere with sterilization efficacy. Instead, they enhance the overall performance of the sterilization process, particularly in high-density instrument loads or complex device configurations where moisture retention is more likely.
Regulatory Requirements: FDA, ISO, and EU MDR Perspectives
From a regulatory standpoint, wet packs are explicitly recognized as a critical risk factor affecting sterility assurance. Under ISO 11607—recognized by the U.S. FDA—sterile barrier systems must maintain sterility after processing and prevent microbial ingress. Residual moisture is considered a key contributor to sterility failure due to its ability to facilitate microbial penetration.
FDA guidance further emphasizes that sterilization packaging systems must undergo comprehensive validation, including verification of drying performance and condensate control. Similarly, the European Union Medical Device Regulation (EU MDR 2017/745) requires that sterile packaging systems maintain sterility throughout storage and transportation and must be designed to minimize contamination risks associated with moisture.
Clinically, wet packs are widely regarded as an indicator of sterility compromise. Once identified, affected instruments must be re-cleaned and re-sterilized. Therefore, implementing moisture control strategies—such as absorbent liners—is consistent with risk management principles outlined in ISO 14971 and is increasingly considered a best practice in compliant CSSD operations.
Compatibility with Specialized Container Designs
Certain sterilization containers incorporate internal metal brackets or fixation structures to stabilize instruments. In such configurations, absorbent liner pads remain highly adaptable. They can be perforated or penetrated to accommodate fixation components without compromising their absorption performance.
This is because the absorption mechanism of these liners relies on capillary action within their fibrous structure rather than on material integrity alone. Even when locally punctured, the overall moisture absorption capacity remains effective. This flexibility allows seamless integration into various container designs without requiring changes to instrument arrangement or workflow.
Clinical Recommendations and Risk Mitigation Strategies
Based on clinical experience and regulatory expectations, absorbent liner pads should be incorporated into standard operating procedures (SOPs) for sterilization container use. Their application is particularly recommended in high-risk scenarios, such as high-density instrument loads, extended sterilization cycles, heavy metal instrument sets, and containers with a history of wet pack occurrences.
Additional risk mitigation strategies should include optimizing sterilizer drying parameters, avoiding overloading that impedes steam circulation, and routinely inspecting container filters and ventilation systems. Through a systematic and standardized approach, healthcare facilities can significantly reduce wet pack incidence and enhance sterilization quality.
Conclusion
The use of absorbent liner pads in sterilization containers is both clinically necessary and aligned with global regulatory standards. By effectively managing residual moisture, they address a key vulnerability in container-based sterilization systems. Their integration into CSSD workflows not only improves compliance with FDA, ISO, and EU MDR requirements but also enhances patient safety, operational efficiency, and overall sterility assurance.